·
Is the obtaining and using metals still on your minds from I/GCSE Chemistry?
Metals
Ores- are rocks which contain compounds of metal in them.
Reactive metals are found as compounds.
Non-reactive metals are found on their own-gold, silver and platinum
Examples of ores:
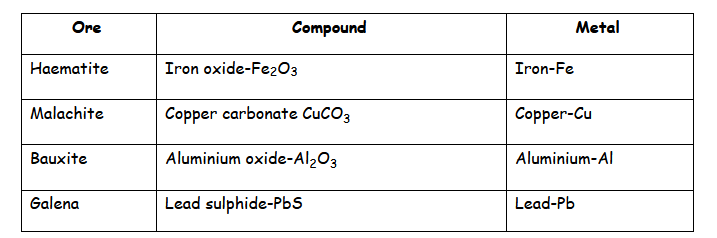
Extraction of Metals:
This means removing whatever element is combined with the metal.
Usually the element is oxygen – so oxygen is removed.
Removal of oxidation is a type of reaction called reduction.
Addition of oxygen is oxidation- e.g burning, rusting
In I/GCSE Chemistry, there are two ways in which a metal can be ‘reduced’ or the oxygen is removed:
1. Heating with carbon
2. Electrolysis

Drafted by Juno Wong(I/GCSE Chemistry)

